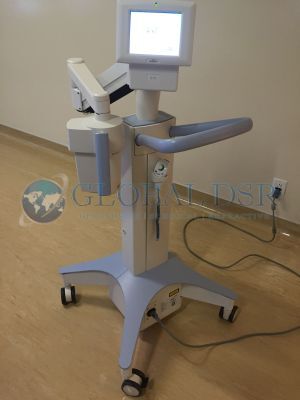
Avedro KXL Sytem Corneal Cross-linking Device
Date of Manufacture 2010
Avedro's KXL System is the only FDA approved cross-linking device:
- UVA Irradiation: 30 minutes at 3mW/cm2
- Laser alignment for patent positioning
- Wireless control for beam alignmentin the X,Y and Z axes
- Fully-integrated stable delivery platform
- Touch screen operation
- Self-calibration of UVA irradiation intensity
Corneal Cross-Linking
- Creates new corneal collagen cross-links
- Results in a shortening and thickening of the collagen fibrils
- Leads to the stiffening of the cornea
Disclaimer:
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. If so, do not purchase this item unless you are an authorized purchaser. If the item is subject to FDA regulation, I will verify your status as an authorized purchaser of this item before shipping of the item. If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA's Center for Devices and Radiological Health: www.fda.gov/cdrh/industry/support/index.html
Return Policy
Items are sold as-is with no returns or refunds available unless explicitly stated.